The Chemistry of Life (Chapter 2)
A. Key Forces Acting on Matter
|
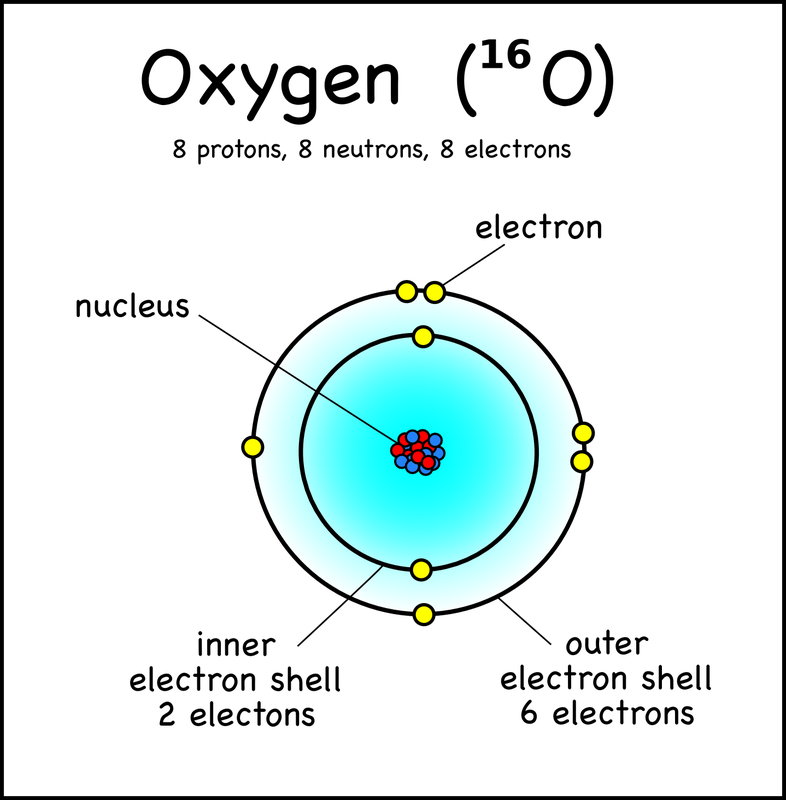
C. The Atom |
B. Matter
|
D. Sub-Atomic Particles
|
E. Chemical BondsDefine in your own words, give examples, include images and sources used to reference for the following bond types (in order of decreasing strength): See Covalent Bonds Colums Below as a Model for All Belo
Covalent Bonds are formed when valence electrons in atomic orbits are shared. Examples of Covalently Bonded Molecules:
Ionic Bonds are formed when valence electrons in atomic orbits are traded. Examples of Ionically Bonded Molecules:
Metallic Bonds are formed between metal atoms when valence electrons form a "metallic electron sea". Examples:
Hydrogen Bonds are... Van der Walls Forces a |
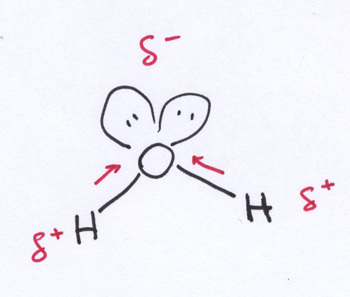
F. Water and It's Importance to Life
Water is a polar molecule because... Adhesion is water's ability to attract to its____. Cohesion is water's ability to attract to other mole_____. Adhesion and cohesion account for water's ability to be able to travel up roots to the leaves of trees since.... (explain further in a couple sentences).
A solute is a substance that... A solvent is a substance that... Water is the universal solvent because... A solution is when the solu___ is uniformly distributed within the solve____. A suspension is a heterogeneous mixture in which not all particles are distributed uniform____.
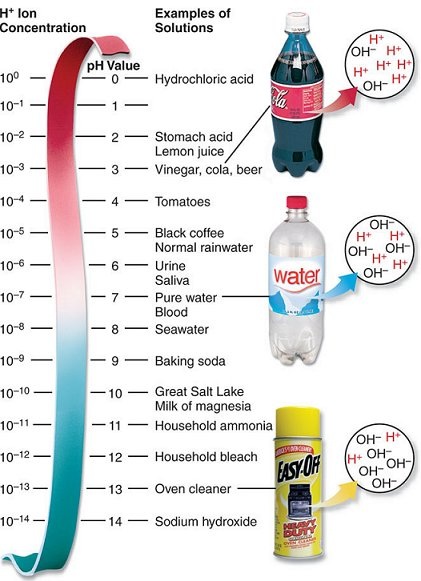
pH is a measure of the amount of hydrogen ions in solution. An acid contains more H+ ions than _____ ions. A base contains more OH- ions than _____ions. |